If you thought that the manufacturing, marketing and sale of nutritional supplements was heavily regulated and closely monitored, think again! In this article, we’ll uncover the hard truths about how supplements are regulated, and how manufacturers can get away with making bold or misleading statements about their products without actually needing to provide any supporting evidence to back-up their claims.
Before we get started, it is first necessary to define exactly what a nutritional supplement is. According to the Food Standards Agency (1), nutritional supplements are described as ‘a concentrated source of a vitamin, mineral, or other substance with a nutritional or physiological effect, alone or in combination and is sold in dose form’.
Nutrition Supplements are Big Business
The nutritional supplement industry was estimated to be worth an astonishing £102.33 billion in 2020 (projected to reach £110.65 billion by 2028) (2). In-fact, the majority of adults in the UK now take a nutritional supplement, with the top reason reported ‘to maintain general health and wellbeing’ (3).
Research shows that supplements are not actually making people healthier (4). Instead, what is actually occurring is that those people that already lead a healthy lifestyle (e.g. those who exercise and who are more conscious about what they eat) tend also to be the ones taking nutritional supplements, making supplements appear health to be health promoting by default (4). In-fact, a recent position statement from the British Medical Journal, concluded (4):
“Current evidence does not support recommending vitamin or fish oil supplements to reduce the risk of non-communicable diseases among populations without clinical nutritional deficiency”
Therefore, unless somebody is consuming extremely low amounts of a particular nutrient (e.g. vitamin C, Omega-3 etc) from their diet, then there is very little benefit to be gained by consuming those nutrients in nutritional supplement form.
In reality, clinical deficiency is very rare in the industrialised Western world, considering higher quality diets, food fortification, and public awareness about health. This begs the question, if the British Medical Journal (one of the most trusted sources of scientific medical information in the world) does not endorse the use of supplements to promote health, why are so many UK adults still spending absurd amounts of money on products they are probably not getting much (if any) benefit from?
Throughout this article we will discuss the reasons that have contributed to this concerning state of affairs, and what could potentially be done to address this in the future. If you are a fitness instructor, personal trainer or nutrition coach, then it’s important that you have a strong understanding of many of the issues presented in this article, so that you can help your clients to make better and more informed decisions about their health. You’ll also likely stop them from literally pouring their money down the toilet.

Is the Supplement Industry Regulated?
The short answer to this question is yes, but not in the way that many people think that supplements are regulated or controlled. A supplement manufacturer cannot make medicinal claims about their products which suggests that their product can prevent, treat or cure a disease or medical condition (e.g. ‘this product cures cancer’). Such a claim must be supported by a substantial amount of scientific evidence and would be subject to close scrutiny and regulation by The Medicines and Healthcare Products Regulatory Agency (MHRA), that is responsible for licensing supplements that make medicinal claims (5).
Considering the MHRA is responsible for ensuring that medicines and medical devices are acceptably safe (e.g. vaccines), the agency has a very high bar for scientific evidence, meaning very few, if any, nutritional supplements would actually meet this standard and subsequently obtain a medicinal claim license. If supplements had been proven effective at fighting disease, they would typically be available on the NHS, like a folic acid supplement is during pregnancy. It’s also worth noting that those companies producing these supplements would also be making trillions of pounds in government contracts and would certainly not need to waste their time or money promoting their products to unsuspecting healthy people.
How Nutritional Supplements Are Actually Regulated?
If a supplement company makes a nutrition or health claim about their product, like ‘maintains healthy cholesterol levels’ for example, then this claim must also be authorised by the UK Nutrition and Health Claims Committee (UKNHCC). The UKNHCC is a scientific advisory committee of eight leading experts in nutrition, health, and sport science (6). The UKNHCC is actually a division of Public Health England.
Companies can directly apply for permission to make a claim about their product, or , if they do make such a claim and they have not sought the appropriate clearance, they may be reported for making a false claim. In both scenarios, the UKNHCC will examine the supporting scientific evidence for the claim and will decide whether the company is permitted to make, or continue making such a claim.
Positively, this means that supplement companies cannot make medicinal, health or nutrition claims that they are not authorised to make without approval from a scientific advisory committee (5). However, in reality, companies can make claims about their products and will only ever be challenged and held to account by the UKNHCC if somebody actually decides to report them. Even when they are reported, there really aren’t any appropriate sanctions or penalties for misrepresenting the benefits of their products.
Supplement manufacturers simply have to convince the consumer that a product is effective. Despite these companies not being legally permitted to make false claims, 73.8% of individuals who consume nutritional supplements still believe that the products they are buying are well researched and supported by evidence (3) when in reality, they are not. This begs the question, why are people still being convinced that nutritional supplements are supported by rigorous and reliable evidence?

Is Supplement Marketing Misleading?
Again, the short answer to this question is ‘yes’. Supplement companies will typically use some form of statement alluding to the fact that a product ‘contributes to the normal functioning of ‘X, Y or Z’. For example, vitamin C products are widely promoted on the premise that they ‘contribute to the normal functioning of the immune system’. Just in case you didn’t spot what they did here, the key words here are ‘contributes to’ and ‘normal functioning’ – they didn’t say ‘causes’ or ‘improved functioning’, as most people would interpret this statement. This careful and meticulous use of language ensures the product advertisement is not in breach of any of the industry regulation standards. Nevertheless, people still automatically associate ‘normal immune function’ with preventing a cold, and ‘normal cholesterol levels’ with reduced cardiovascular disease risk.
Supplement marketing also involves a bait and switch. An advertisement will often hook a viewer in with a common sensation of everyday life, like tiredness for example, alongside an image of the supplement or its packaging. Consequently, the unsuspecting customer’s brain fills in the gaps, again, ensuring that the manufacturer isn’t actually in direct breach of any regulatory standards.
Supplement manufacturers don’t just rely on their customer’s naivety, they also utilise indirect marketing tactics to drive sales. Most supplement manufacturers operate blogs and websites (similar to this one), where various health conditions, diseases, and performance-based subjects are covered. After presenting the usual lifestyle advice (exercise, healthy diet, manage stress, sleep, etc), these content pieces will conveniently make reference to the use of a particular supplement as an option to be used alongside the other strategies presented. The internet is literally laden with low-grade content, typically written by laymen and copywriters, tasked with writing about all manner of ailments and conditions, where links to the ‘potential benefits’ of these supplements can be made.
These articles and blogs are technically not in breach of any of the industry regulation because they never actually state that supplement reduces the risk of a disease, or medical condition. However, the content of these articles is usually very questionable from an ethical and scientific perspective because it will strongly imply that the focus condition or disease may actually be improved by using a particular nutritional supplement.
Finally, many of these articles and blogs are published on third-party websites and written by freelance copywriters, so there are no outward or direct links to the company or product being featured. This again circumvents any potential clash with industry regulators but still ensures that the right amount of misinformation and confusion is created when consumers consult with ‘Doctor Google’ for their particular ailment or symptom.
There are literally tens of thousands of websites, social media campaigns, YouTube videos, and podcasts on the internet that significantly overstate the benefits that nutritional supplements have on health. These platforms have their own marketing agenda, meaning the more sensational the headline is (e.g. top 10 supplements to fight cancer), the more clicks, views, and subscriptions, the platform will receive. Very often, many of these websites also generate colossal revenues from display advertising (the more people read the content, the more money they generate from ads).
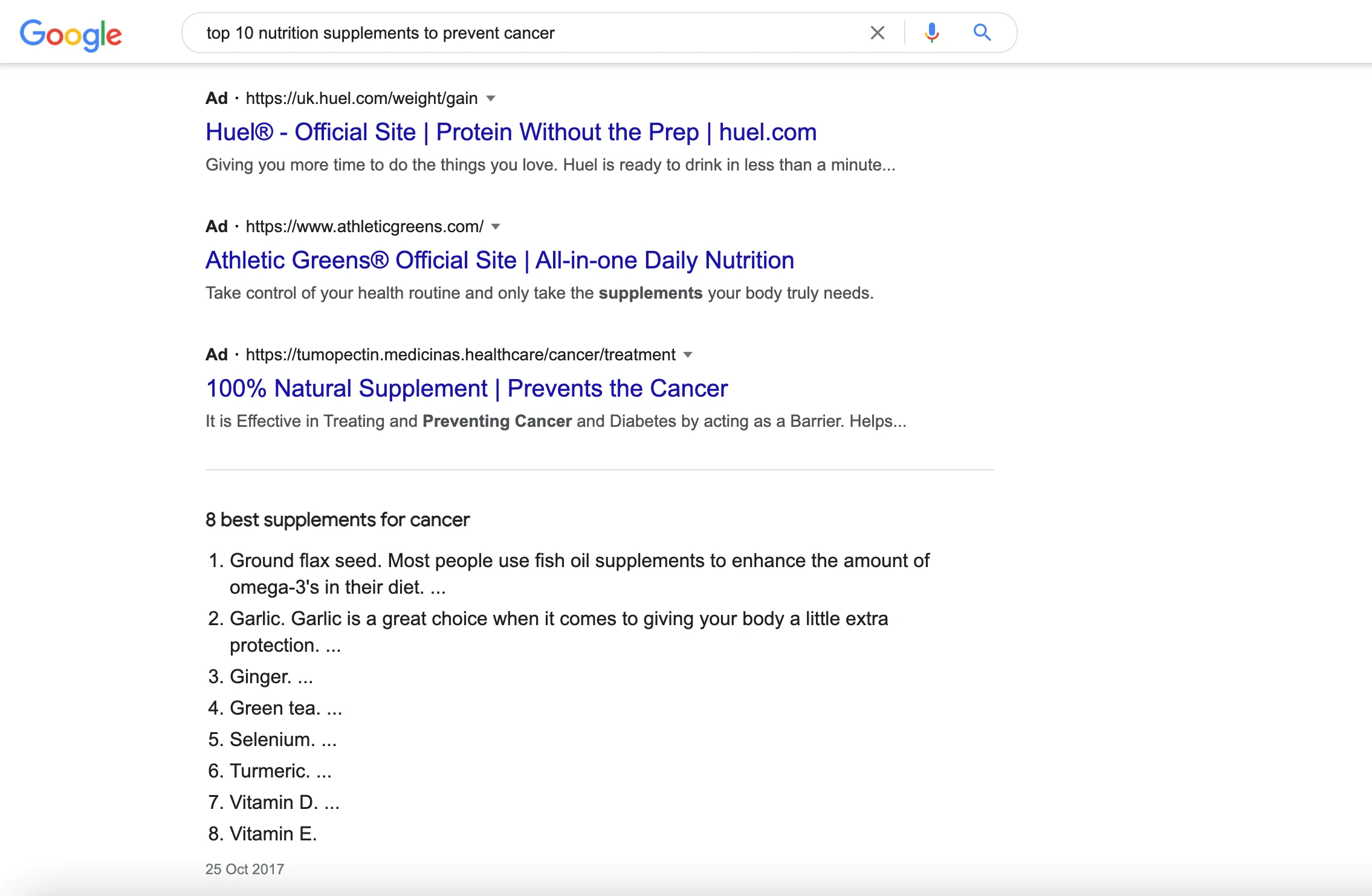
The Spread of Supplement Misinformation
Unfortunately, sensationalist headlines cause misinformation to spread like wildfire, and uphold the falsehood that supplements directly improve health.
In the age of smartphones, social media, and hyper-connectivity, such misinformation is now more accessible than ever before. Misinformation translates into increased sales for supplement companies. In-fact, the top two reasons why people say they are convinced to purchase a nutritional supplement is ‘personal research’ (38.1%), or a ‘family/ friend recommendation’ (27.6%) (3).
Unfortunately, there is a lack of appetite to stop the spread of misinformation on the internet. There are no actual laws that prevent misinformation online because this is seen to be preventing people’s freedom of expression (7). Ultimately, people want to believe supplements improve health, perhaps because taking a daily tablet is much easier to achieve than changing old habits.
Online platforms want more subscribers and supplement companies want to sell more products, so it’s in the interest of both parties to ensure that the spread of misinformation perpetuates. It would certainly be useful to consumers if supplement manufacturers were made to be more accountable for their actions and statements, either explicit or implied. Perhaps Government agencies could take stronger actions, including issuing hefty fines, for those companies that are found to be misleading their customers? However, unlike other food products that are sold in shops (e.g. bread, fruit, vegetables etc), nutritional supplements are actually ‘in scope’ for VAT, and are charged at the standard rate of 20%. Given that we’ve already established that the size of the supplement business is literally colossal, the UK Government actually benefits from unethical and misleading tactics employed by these companies. Quite literally, the UK Government actually received millions in tax receipts from the sale of nutritional supplements and is perhaps in part why there is little impetus to change the current regulatory infrastructure.
Conclusion and Recommendations
To summarise, current evidence suggests that nutritional supplements do not improve health. Despite there being clear regulation preventing a manufacturer from making false claims, people are still being grossly mislead and duped in to spending absurd amounts of money on nutritional supplements. This is achieved through a combination of clever marketing, unethical advertising, and the wide spread of online misinformation. There appears to be a general lack of desire or interest in regulating online misinformation, and so the responsibility is now on exercise professionals to ensure that their clients receive accurate and reliable information. We must question whether a supplement really is effective before buying it, and better educate ourselves and our clients on how to spot misinformation online. Finally, we must call out companies who are breaching industry regulation standards and deliberately misleading people for simple profit.
You can report a company making false claims about their products to: [email protected]
Interesting in learning more about nutrition supplements and their efficacy? Why not consider our nutrition supplements CPD course designed for personal trainers and qualified nutrition coaches. Alternatively, you may want to specialise in nutrition coaching, in which case you may be interested in completing our nutrition and weight management coaching courses. All our programmes are developed and delivered by leading experts with decades of industry experience.
References & Sources
1. Food Standards Agency. (2021). Food supplements. Available at URL: https://www.food.gov.uk/business-guidance/food-supplements (Accessed 04/11/2021).
2. Grand View Research. (2021). Dietary Supplements Market Size, Share & Trends Analysis Report By Ingredient (Vitamins, Proteins & Amino Acids), By Form, By Application, By End User, By Distribution Channel, And Segment Forecasts, 2021-2028. Available at URL: https://www.grandviewresearch.com/industry-analysis/dietary-supplements-market (Accessed 04/11/2021).
3. Health and Food Manufacturers Association. (2019). Health of the Nation Survey (2019). Available at URL: https://hfma.co.uk/wp-content/uploads/2020/07/1107415-hfma-health-of-the-nation-survey-report_v5-1.pdf (Accessed 04/11/2021).
4. Zhang, F.F., Barr, S.I., McNulty, H., Li, D. and Blumberg, J.B., 2020. Health effects of vitamin and mineral supplements. bmj, 369.
5. Department of Health. Food Supplements. Available at URL: https://www.gov.uk/government/publications/food-supplements-guidance-and-faqs (Accessed 04/11/2021).
6. UK Government. UK Nutrition and Health Claims Committee. Available at URL: https://www.gov.uk/government/groups/uk-nutrition-and-health-claims-committee (Accessed 04/11/2021).
7. Helm, R.K. and Nasu, H., 2021. Regulatory responses to ‘fake news’ and freedom of expression: Normative and empirical evaluation. Human Rights Law Review, 21(2), pp.302-328.
